What Makes Wood Heavy?
Chemical composition of plants, scientific reasoning, & misconceptions
Overview
This article reflects on how misconceptions are difficult to change with respect to the chemical composition of wood. It is a summary from the discussion of Video 2 - Lessons From Thin Air 56:24 in the Harvard Smithsonian series: Minds of Our Own trilogy. See related notes
Or to use with science learners who aren't preparing to be teachers and might not be interested in the Minds of Our Own videos might be interested in thinking about the comic from both a poetic and scientific point of view.

Poetic ... your on your own ...
Scientific, use the discussion below.
Enjoy!
Discussion
How deep should a person understand photosynthesis?
What is sufficient to be considered scientifically literate?
Would it be possible to be scientifically literate and not instantaneously realize a log is created from water and carbon dioxide - and it is a combination of hydrogen, oxygen, and carbon that make almost all of its mass?
This article explains how a scientific literate person might come to realize what elements contribute to the majority of the mass in a log and how it is achieved.
First, it is interesting, to think about how an instantaneous response might be given.
Instantaneous in this case would have to be defined as after the questioning event. It could not be simultaneous or before the question was finished being asked as the question needs to be communicated. Additionally some time must elapse after the question is asked for a person to formulate an answer.
Therefore, it would not be possible to give an answer simultaneous with the asking of the question. The question would have to be asked and comprehended before it could be answered.
So time would elapse before an answer was given. How much time must elapse before an answer would be considered beyond a point of spontaneous?
If a person would be able to answer the question within seconds of the question being asked? Would that be spontaneous? I would think so.
What is the likelihood of a person doing so if they never previously answered or heard an answer to the question?
If they hadn't, they would have needed time to recall sufficient information and organize it through thoughtful logical reasoning to arrive at scientifically acceptable answers. It is because of this I believe it is extreemly unlikely a learner would do so within seconds of the question being asked.
If instantaneous extends into minutes and hours, then I would say a scientifically literate person should be able to reason their way to scientifically acceptable answers within that kind of time frame.
You may wonder: why I am making such a big deal out of this.
It is because most people don't realize that when people give quick answers to such questions, they are not creating answers on the spot. The question was presented to them at some previous time and someone or they themselves took ample time to present a convincing argument to support an answer.
The key, for me, is the ability to reason scientifically, not the amount of time taken to reach an acceptable answer. So what information would most people need to know and what ways might they reason with that information to arrive at acceptable answers to be considered scientifically literate?
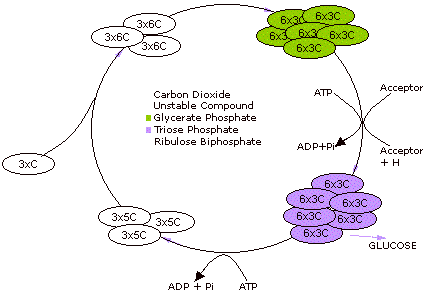
A scientifically literate person would understand photosynthesis and that it involves water, carbon dioxide, and sunlight. In addition they would know it is a reaction that combines carbon dioxide and water with sun energy in a way that converts them to simple sugar and oxygen. In addition they could recall that glucose or simple sugar has the chemical formula of C6 H12 O6 and that six carbon dioxide molecules and six water molecules can create one simple sugar molecule and 6 oxygen molecules.
This information should suggest the answer involves those three elements.
However, a lack of non-authentic learning experiences might inhibit a person's ability to focus on those elements as a necessary part of the answer. A person with a misconception that trees get their food from dirt might focus on dirt, that it is heavy, and reason that dirt stuff makes wood, therefore, the weight of wood comes from the dirt. This focus (centering) on soil overwhelmed their thinking so they did not consider the ingredients used by trees to make wood as being simple sugar (glucose - C6 H12 O6 ) created from air (CO2) and water (H20) photosynthesis.
However, knowing this isn't sufficient to reach a definitive answer. Many people gave inaccurate answers or claimed to not know, probably knew the basic information that could be used to reason with and arrive at an answer. However, they did not believe they had the ability to use logical reasoning with scientific information or could access scientific information needed to use logical reasoning and create acceptable answers. It is essential to understand the nature of science and have enough self-efficacy to believe you can reason in the manner of scientists. If a person has, they will know they must use ideas that include verifiable observable information which has or can be observed repeatedly and combine them with logical reasoning to draw conclusions so they as well as others can understand and verify.
Reasoning from the idea that the basic building block of all plants is - fructose or glucose or simple sugar with the chemical formula of C6 H12 O6, one wonders what the proportion of these elements are in wood and if other elements are involved.
Does sugar really become wood?
If so, how?
If there is more hydrogen in a sugar molecule, is wood mostly hydrogen?
If so, how much?
Might there not be other molecules introduced in the process of making wood?
Further, information in the periodic table of elements includes the fact that hydrogen has an approximate atomic mass of one, carbon - six, and oxygen - eight.
Does that information suggests that one of these three (oxygen) might provide more mass with less atoms?
This seems to suggest that mass will be dependent on the proportion of atoms in the wood more so than the total number of atoms?
These questions spark a desire to want to know the chemical make-up of sugar and wood. Maybe that will provide evidence for an accurate answer. Diagrams are provided following the article so that you can create your own reasonable answers.
Another complication is when we think of oxygen we think of it as a gas. We center on how little it weighs in air. Likewise with hydrogen. However, when these elements are in wood they are in a solid state. Considerably more dense than in a gaseous state.
So how deep does ones understanding have to be?
What needs to happen to create strong enough connections of science ideas so learners can connect ideas (photosynthesis, simple sugar, density of gases, density of solids) to explain such natural occurrences rather than center on less significant but more common sense kind of information related to the ideas (dirt)?
What is needed is to provide learning in science to achieve a desire and acceptable use of scientific reasoning?
What experiences are necessary for students to become aware of their preconceived notions?
What experiences are necessary for learners to replace their preconceived notions with more scientific ideas?
And more importantly the attitudes to systematically study something for the purpose of understanding it.
Science includes many dimensions and many concepts within those dimensions.
How do learners gain access to sufficient dimensional ideas to become scientifically literate?
How do we teach when they already have a set of ideas about why certain events happen in the manner they do?
How do we answer learners' questions when they are not developmentally ready for a scientific answer without adding to their misconceptions?
Yes I know. So many questions.
I suppose the biggest impact this video had on me is the notion that teachers need to convince themselves and others that the answers students give are important. Too often we act as if the only information we need to know about their answer is if they are: right or wrong.
More important, is why they believe their answers are accurate, what information they are using, and the reasoning they use to arrive at their answer. All this implies knowing, explaining, and communicating observational experiences with their reasonings used to conclude their ideas.
Science literacy is more than facts. It includes connections which force our minds to ponder how facts are related in a reasonable understandable predictable way. As Professor Philip Sadler stated in the video, “Science is predicting, not explaining”. And more importantly an attitude to want to be able to predict.
Information about chemical composition of plants.
Chemical composition of three sugars:
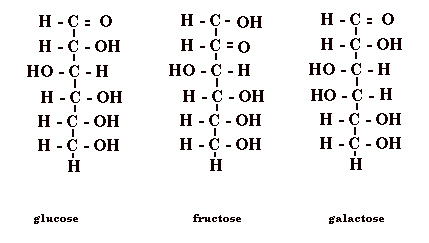
Wood is composed of - cellulose (40%–50%) and hemicellulose (15%–25%) impregnated with lignin (15%–30%)
Chemical composition of cellulose:
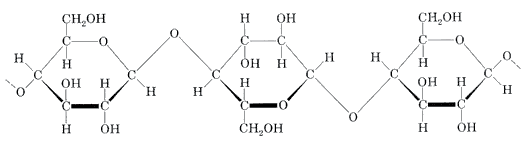
Hemicelluloses are imbedded in the cell walls of plants, sometimes in chains. They bind with pectin to cellulose to form a network of cross-linked fibres.
A hemicellulose can be any of several polysaccharides present in almost all cell walls with cellulose. Hemicellulose contains many different sugar monomers. In contrast, cellulose contains only anhydrous glucose. For instance, besides glucose, sugar monomers in hemicellulose can include xylose, mannose, galactose, rhamnose, and arabinose. Hemicelluloses contain most of the D-pentose sugars, and occasionally small amounts of L-sugars as well. Xylose is always the sugar monomer present in the largest amount, but mannuronic acid and galacturonic acid also tend to be present. While cellulose is crystalline, strong, and resistant to hydrolysis, hemicellulose has a random, amorphous structure with little strength. It is easily hydrolyzed by dilute acid or base as well as myriad hemicellulase enzymes.
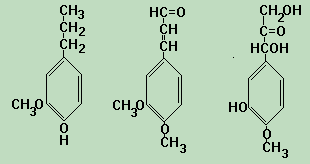
Lignin is in all vascular plants, mostly between the cells, but also within the cells, and in the cell walls. It is the fiber that makes vegetables firm and crunchy. It reinforces the cell walls and vessels that transport liquids by keeping them open so liquids can flow in cells and throughout the plant. It enables plants, particularly trees to grow taller and compete for sunshine. This flow is a disposal mechanism for metabolic wastes as well as source mechanism for essential resources. It is formed when water is removed from sugars and create aromatic structures. These reactions are not reversible. There are many possible monomers of lignin, and the types and proportions depend on the source in nature. Some typical monomers are shown in the sketch
Carbon fixing reactions of photosynthesis
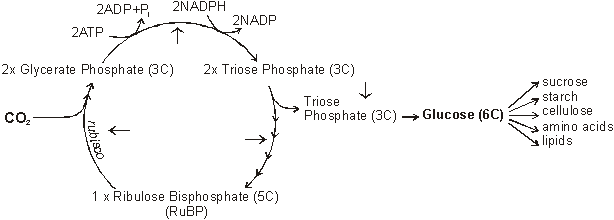
Create:
- sucrose - table sugar
- starch - C6H10O5
- cellulose - see above
- amino acid - a basic amino group (NH2), an acidic carboxyl group (COOH), and an organic R group (or side chain)
- lipids -